Home
m1medical.com
919
601 9053
Home
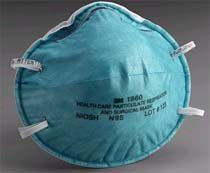
3m Medical Surgical Face Mask 1860 / N95
Respirator - Sold Out
| Item # | 3m Medical 1860 Surgical Mask |
| Item Desc. | |
| List Price | Sold Out |
3M™ 1860 Health Care N95 Particulate Respirator and Surgical Mask.
3M™ 1860 Health Care N95 Particulate Respirator and Surgical Mask.
NIOSH approved as a Type N95 respirator. It meets CDC guidelines for TB exposure
control and is designed specifically for use in a health care setting. It has a
soft inner shell for greater comfort against the face and, because it's fluid
resistant, it aids in reducing the potential exposure of the wearer to blood and
other fluids.
* NIOSH approved as a Type N95 respirator to meet OSHA requirements and CDC
guidelines for TB exposure control.
* Helps reduce wearer exposure to airborne particles.
* >99% BFE1 to help reduce patient contamination caused by exhaled
microorganisms.
* Fluid resistant to help reduce potential exposure of the wearer to the spray,
spatter and aerosol of blood and body fluids.
* Does not contain either natural rubber latex or dry natural rubber as
components in the product or its packaging.
* Respirator includes a soft inner shell for greater comfort against the face.
* Hypoallergenic to reduce chance of allergic reaction.
* Individually packaged in zip lock bag.
* Instructions included.
* 1 - Bacterial Filtration Efficiency (BFE) determined by the modified Greene
and Vesley test method.
| Item # | 3m 1860 |
| Item Desc. | N95 Respirator and Surgical Mask |
| List Price |